Unlocking the Scientific Subgroup: Pressnies Inhibitor Gastr Hoy Skin Identifiable
As the human body continues to mystify us with its intricate mechanisms and complex interactions, researchers delve deeper into the realms of biomedical literature to unravel the mysteries of various conditions and treatments. The intricacies of gastric acid secretion, motility, and the various proteins involved in these processes are of particular interest to those studying the gastrointestinal tract. At the forefront of this inquiry are the proton pump inhibitors (PPIs), a class of medications used to treat acid-induced disorders and conditions such as gastroesophageal reflux disease (GERD).
What are Proton Pump Inhibitors (PPI)?
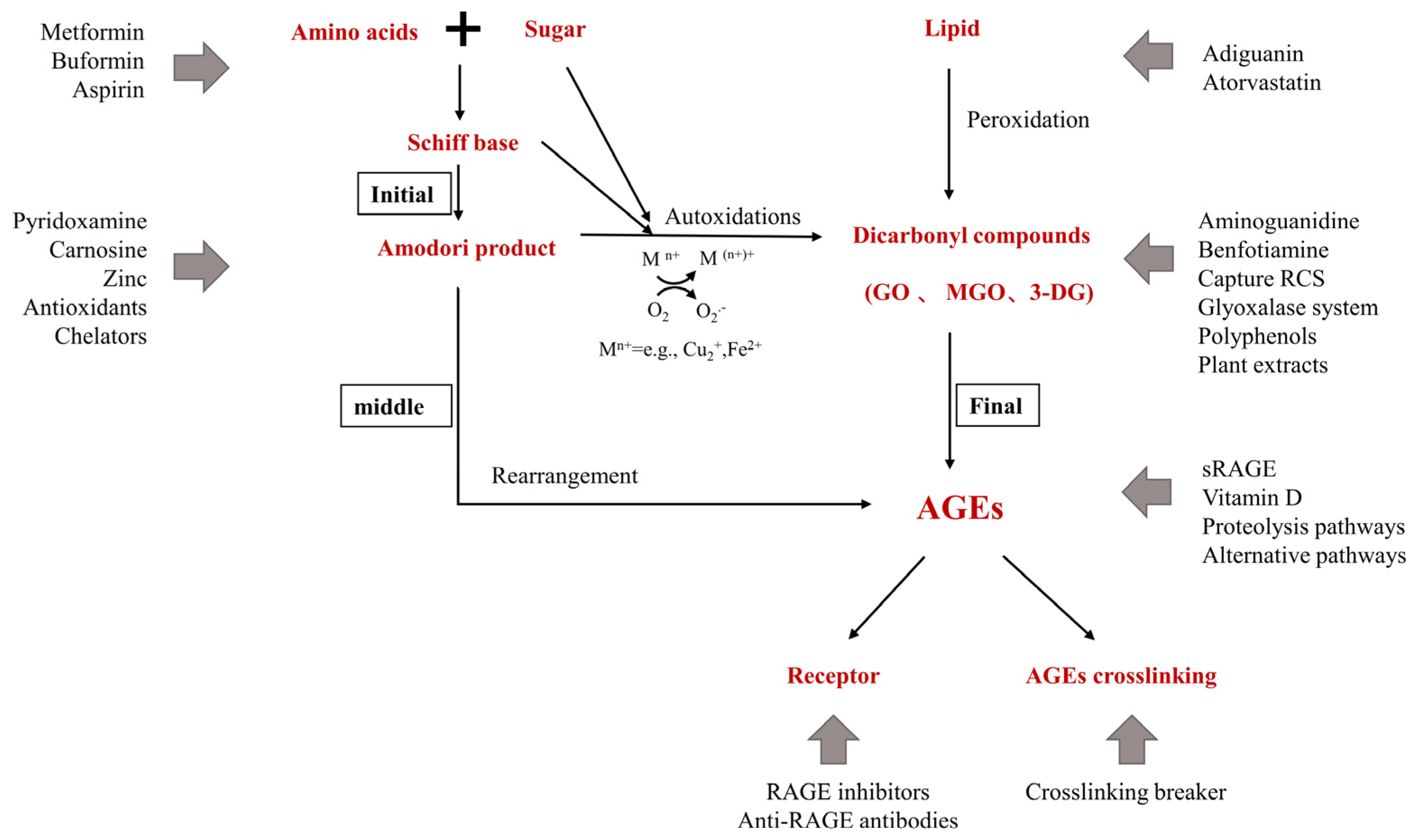
Proton pump inhibitors, or PPIs, are a group of drugs that inhibit gastric acid secretion by blocking the H/K ATPase pump within the parietal cells of the stomach. These pumps are crucial in the generation of stomach acid, and by inhibiting them, PPIs significantly reduce gastric acid secretion. This mechanism allows PPIs to alleviate symptoms associated with excessive gastric acid, such as heartburn and acid reflux.
Pressnies Inhibitor Gastr Hoy Skin Identifiable: A Scientific Subgroup
Deep within the vast expanse of biomedical literature and scientific databases, such as PubMed, lies a specific subgroup of individuals who have been studied in relation to gastrointestinal disorders. This cohort includes those who have used PPIs and have experienced skin reactions, including angioedema and urticaria, which are more frequent than anaphylaxis. The concept of "pressnies inhibitor gastr hoy skin identifiable scientific subgroup" refers to a subset of individuals within this broader group, characterized by their gastrointestinal symptoms and adverse reactions to PPIs.
Pathophysiology and Treatment of Gastric Hypersecretion and Motility
Eliminating or reducing gastric acid secretion can significantly impact treatment outcomes in conditions involving hypersecretion, such as Peptic Ulcer Disease, Helicobacter pylori eradication, and it is covered with the somatostatin analogs such as octreotide. The parietal cell secretory process holds significant relevance in the management of such conditions. Adopting the correct medication strategy is essential in these treatments as they significantly meter the secretion of stomach acid.
Side Effects of PPIs: An Examination of Skin Reactions

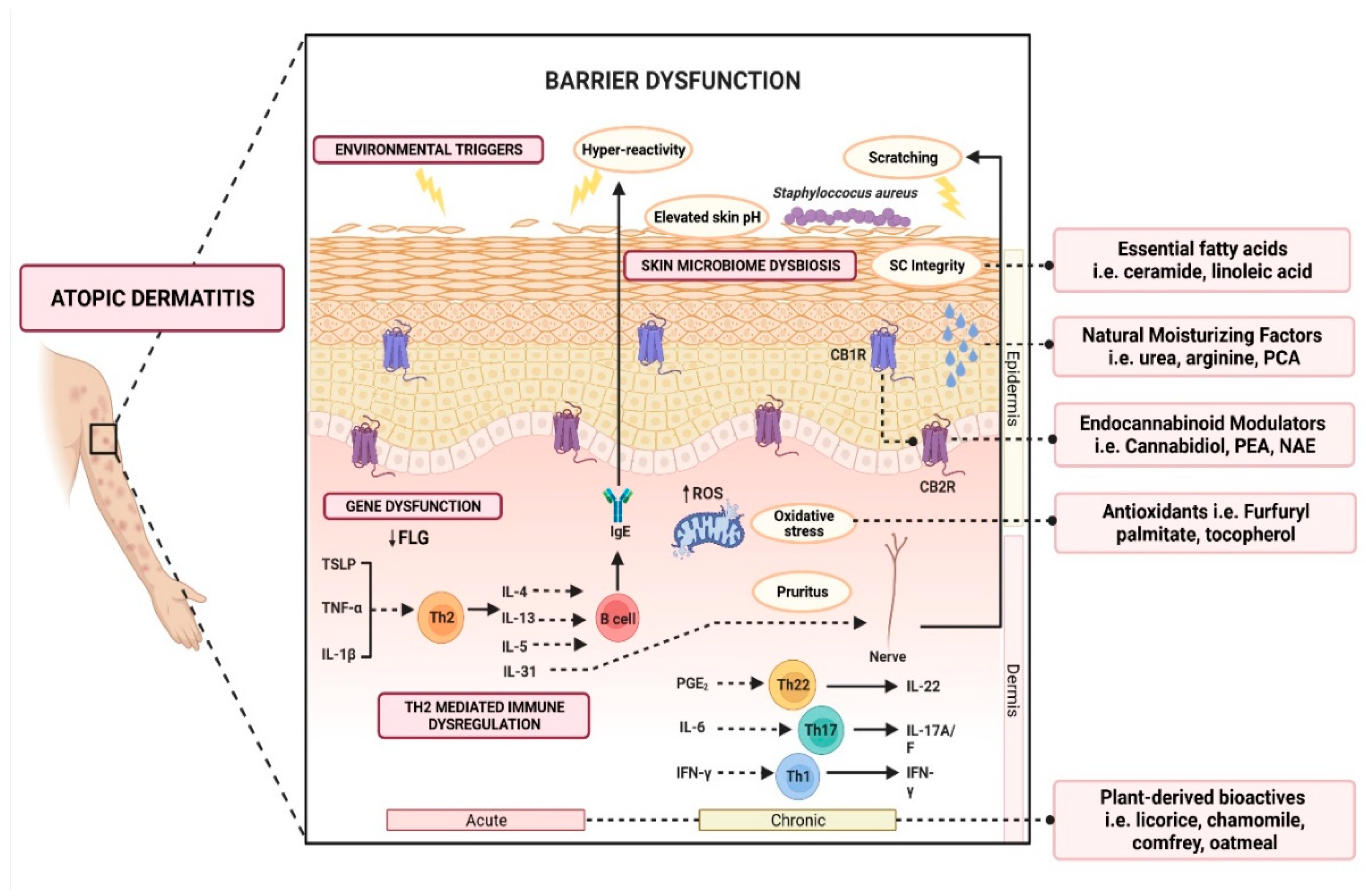
While PPIs are generally well-tolerated, skin reactions can occur, albeit rarely. Among the documented adverse events, angioedema and urticaria are the most prevalent manifestations. Approximately 0.24% of cases may experience anaphylaxis, a severe and potentially life-threatening allergic reaction. Understanding these skin reactions is indispensable for researchers examining the effects of PPI-induced gastrointestinal tract conditions.
Long-Term Use of PPIs: Risks and Considerations
The widespread use of PPIs for acid-related disorders extends beyond the scope of treating immediate symptoms. Long-term use can be associated with various risks, including an increased risk of psoriasis, especially in individuals with
Conclusion: The Complex Relationship Between PPIs and Skin Reactions
The pressnies inhibitor gastr hoy skin identifiable scientific subgroup represents a subset of patients within a broader population affected by gastrointestinal disorders and the drugs used to treat them. Understanding the effects and adverse reactions associated with PPI therapy continues to be an area of active research, providing essential insights into the complex interplay between medication, body, and disease.
Recommendations for Future Research
Further studies are imperative to elucidate the long-term effects of PPI therapy on gastrointestinal function, the risk of psoriasis in relation to PPI use, and the molecular mechanisms behind skin reactions and anaphylaxis. These investigations will shed more light on managing gastrointestinal disorders effectively, while minimizing the risk of adverse reactions.